The prevalence of hyperthyroidism in pregnancy in the United States is 0.1 to 0.4%,with Graves’ disease as the etiology of 85% of cases.1,2 Asian women are more likely to have gestational thyrotoxicosis and hyperemesis gravidarum.3 Genetic factors are believed to affect the production and metabolism of human chorionic gonadotropin (hCG) in Asian women. Pregnancy is a very dynamic condition, with manifestations that may mimic some hyperthyroid symptoms. Dysfunctions of the maternal thyroid gland can affect the fetus particularly during the first trimester, due to the inability of the fetus to produce its own thyroid hormone. Hyperthyroidism caused by Graves’ disease may enter remission during pregnancy and then relapse after labor. Untreated hyperthyroidism during pregnancy increases the risk of preeclampsia, heart failure, thyroid crisis, and maternal mortality. 2,4,5
The prevalence of hypothyroidism during pregnancy is 0.3 to 0.7%.4 There are no available data for cases in Indonesia, as screening for thyroid dysfunction is not universally performed.6,7 Autoimmune thyroid disease (Hashimoto’s thyroiditis) and ablation from radioactive iodine therapy are the two most common etiologies; oversuppression of hyperthyroidism is also an important cause. Untreated hypothyroidism during pregnancy also increases maternal and fetal morbidity. Mothers with severe hypothyroidism may have anemia, myopathy, congestive heart failure, preeclampsia, placental anomalies and postpartum hemorrhage. Undetected congenital hypothyroidism can cause developmental, cognitive and neurological damage.5,8,9
The guidelines on thyroid dysfunction during pregnancy were developed by the Indonesian Society of Endocrinology.
Click here to download Table 1
Table 1. Thyroid function in normal pregnant and nonpregnant women11
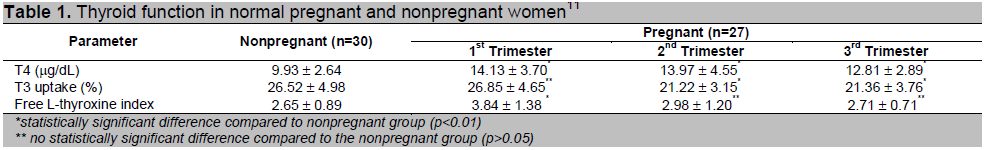
The diagnosis of hyperthyroidism in pregnancy should be based on the patient’s history, physical findings and laboratory examinations, bearing in mind that thyroid scintigraphy is contraindicated during pregnancy.1,2,4,5,6,10
Hyperthyroidism in pregnancy might present as incongruous weight loss or gain during pregnancy, ophthalmopathy, unidentified struma, muscle weakness, onycholysis, and rarely, pretibial myxedema.5 Weight loss in hyperthyroidism is confounded by weight increase during pregnancy. Physical findings in hyperthyroidism such as increased cardiac output, systolic murmur, tachycardia, warm skin and heat intolerance are also found in normal pregnancy. A stable resting heart rate of 100 beats per minute during Valsalva maneuver suggests the diagnosis of thyrotoxicosis.6,10
Testing for thyroid stimulating hormone (TSH), free thyroxine (FT4) and thyroid receptor antibody (TRAb) concentration should be done in pregnant women suspected to have hyperthyroidism. It is important to consider the physiologic effects of hCG, causing a lower TSH level and a higher thyroxine-binding globulin (TBG) concentration during pregnancy. TSH concentration at the end of the first trimester may reach a level of 0.03 mIU/mL in normal pregnancy. As such, a solitary low TSH level is not always indicative of hyperthyroidism. Physiologic TBG elevation during pregnancy can affect the proportion of circulating total thyroid hormone, making free thyroid hormone testing necessary.1,2,4-6,8,10
Two types of thyroid antibodies are known to be elevated in Graves’ disease: thyroid anti-microsomal antibodies (thyroid peroxidase antibodies, TPOAb) and thyroid stimulating hormone receptor antibodies (TRAb). Thyroid-stimulating immunoglobulin (TSI) and thyroid binding inhibitory immunoglobulin (TBII) are two of the TRAbs that may be tested. TRAbs have better prognostic value for the development of hyperthyroidism in the fetus. These antibodies are found in almost all conditions of Graves’ disease but not in gestational thyrotoxicosis. Laboratory tests may be reevaluated at 3- to 4-week intervals. 4,5,8
TRAb examination is recommended for mothers with a history of Graves’ disease, previous treatment with radioactive iodine or surgery before pregnancy, or those with children diagnosed with Graves’ disease. The tests should be done at the beginning of the pregnancy and repeated in the last trimester. High concentration of TRAbs before labor indicates thyroid dysfunction in the fetus.4,8
Anti-thyroid medication is the first-line therapy for hyperthyroidism during pregnancy. Surgery may be indicated in select cases, while iodine-131 (I-131) therapy is contraindicated during pregnancy.8,10
The goal of anti-thyroid treatment is to restore maternal thyroid function, which may or may not affect fetal thyroid function.1,2,4-6,8,10 Recent studies and reports showed that propylthiouracil (PTU) or methimazole (MMI) can be given safely during pregnancy. Adverse events that may occur include as agranulocytosis, liver dysfunction and congenital anomaly (MMI).The dosage of anti-thyroid medication during pregnancy should be adjusted to maintain the lowest dose to achieve maternal euthyroidism and to avoid fetal hypothyroidism. Anti-thyroid treatment has to be monitored and titrated according to the mother’s thyroid function.1,2,4-6,8,10 FT4 examination is recommended every 2 to 4 weeks to aid dose adjustment and decrease the risk of maternal and fetal hypothyroidism. The target FT4 within the upper third of the normal range for non-pregnant women is considered safe in preventing hypothyroidism in the fetus. Within this recommended range, 90% of fetuses will have normal concentrations of FT4. Maternal FT4 concentration below the normal range causes fetal FT4 to decrease, conferring a higher risk of hypothyroidism to the fetus.8,10
More than 50% of pregnant women with hyperthyroidism will not need an anti-thyroid medication until after the end of the second trimester to the time of labor. Nevertheless, relapse may occur at 3 to 6 months post partum.8,10
Pregnant women diagnosed with hyperthyroidism may be given PTU 100 to 150 mg every 8 hours, or methimazole 10 to 20 mg in single or divided doses (every 12 hours). When the thyrotoxic condition has been controlled—indicated by improvement of symptoms, increase in TSH to normal and decrease in FT4—PTU may be tapered to 50 mg given at 2 to 3 times daily, until the minimum maintenance dose of 50 mg per day; or methimazole may be decreased until the minimum dose of 2.5 to 5 mg a day. Administration of PTU at 300 mg daily is considered safe without causing hypothyroidism to the fetus. If hyperthyroidism persists clinically and biochemically, PTU may be tapered to 100 mg daily until the last 4 weeks before labor 8,10
Propranolol may also be used as an option to control symptoms of acute hyperthyroidism and preoperative management in pregnancy. Long term administration is not recommended. Developmental dysfunction as a side effect of propranolol is still controversial, but hypoglycemia, apnea and bradycardia may resolve in 48 hours. Acute thyrotoxicosis can be controlled by administering propranolol 20 to 40 mg every 6 hours, or atenolol 50 to 100 mg daily. Beta-blockers may be stopped once the target heart rate of 100 beats per minute or less has been achieved.1,2,4-6,8,10
Radioactive iodine therapy is contraindicated during pregnancy because it can cause fetal thyroid dysfunction, neural damage or congenital anomalies. If anti-thyroid medication is given in high doses for an unsupervised period and is found to cause adverse effects, surgery should be considered. Thyroidectomy during pregnancy may be performed safely during the second trimester, mainly due to complete fetal organogenesis and relative stability of the uterus to the contractile effect of medications needed in surgery.2,5,8
It is not recommended to give anti-thyroid medications in cases of subclinical hyperthyroidism (low TSH, normal FT4 and T4 levels without specific hyperthyroid symptoms) during pregnancy due to lack of data on fetal effects. Careful observation is considered sufficient.5,8
The diagnosis is established based on symptoms such as fatigue, excessive weight gain, goiter, constipation and low heart rate. 1,2,5,8
Experts recommend evaluation of TSH concentration in high risk women even before pregnancy, particularly those with known past or family history of thyroid dysfunction and goiter.6-9 Thyroid function tests reveal elevated TSH and low FT4 levels.1,2,5,8 Elevated TPOAb and anti-thyroglobulin antibody (TgAb) levels are common findings in Hashimoto’s thyroiditis, and are recommended for the diagnosis.8
During the early weeks of pregnancy (8 to 16 weeks), levothyroxine replacement needs to increase by 25 to 50%.12-15 After delivery, levothyroxine dose should be lowered to the pre-pregnancy dose. TSH testing must be done every 4 weeks during therapy.
Levothyroxine should be immediately started once the diagnosis is made. In the absence of heart failure, the dose of levothyroxine should be started from 100 mcg daily for 3 to 5 weeks, and then subsequently titrated according to serial TSH exam every 4 weeks until TSH falls within target range. Additional 50 mcg may be given if the desired TSH is not yet achieved. Because thyroid hormone absorption is impaired by iron-containing pregnancy supplements, levothyroxine should be taken at least 2 to 3 hours after iron supplementation. 2,8,12
References
1. Glinoer D. The regulation of thyroid function in pregnancy: Pathways of endocrine adaptation from physiology to pathology. Endocr Rev 1997;18(3): 404-33.
2. Kariadi SHKS. Disfungsitiroidpadakehamilan. Dalam: Djokomoeljanto R, ed. Buku Ajar Tiroidologi Klinik. Balai Penerbit Universitas Diponegoro, Semarang, 2007.
3. Price A, Davies R, Heller R et al. Asian women are at increased risk of gestational thyrotoxicosis. J Clin Endocrinol Metab 1996;81(3):1160-3.
4. Burrow GN. Thyroid disease. In: Burrow GN, Ferris TF, eds. Medical complications during pregnancy. 4th ed. W.B. Saunders Company, Philadelphia, 1995.
5. Fantz CR, Dagogo-Jack S, Ladenson JH et al. Thyroid function during pregnancy. Clin Chem 1999;45(12):2250-8.
6. Lazarus JH, Premawardhana LD. Screening for thyroid disease in pregnancy. J Clin Pathol 2005;58(5):449-52.
7. Vaidya B, Anthony S, Bilous M et al. Detection of thyroid dysfunction in early pregnancy: Universal screening or targeted high-risk case finding? J Clin Endocrinol Metab 2007;92(1):203-7.
8. Abalovich M, Amino N, Barbour LA et al. Management of thyroid dysfunction during pregnancy and postpartum: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab 2007;92(8 Suppl):S1-47.
9. Kempers MJ, van Tijn DA, van Trotsenburg AS et al. Central congenital hypothyroidism due to gestational hyperthyroidism: Detection where prevention failed. J Clin Endocrinol Metab 2003;88(12):5851-7.
10. Shankar P, Kilvert A, Fox C. Changing thyroid status related to pregnancy. Postgrad Med J 2001; 77(911): 591-2.
11. Hirawan Supran. Kadar T4, ambilan T3 danindek stiroksinbebaspadawanita hamilsehattiap semester di RSCM. Tesis, Departemen Ilmu Penyakit Dalam, FKUI Jakarta, 1984.
12. Nikfar S, Koren G. Managing hypothyroidism during pregnancy. Can Fam Physician 2001;47:1555-6.
13. Toft A. Increased levothyroxine requirements in pregnancy—Why, when, and how much? N Engl J Med 2004;351(3):292-4.
14. Tamaki H, Amino N, Takeoka K et al. Thyroxine requirement during pregnancy for replacement therapy of hypothyroidism. Obstet Gynecol 1990;76(2):230-3.
15. Alexander EK, Marqusee E, Lawrence J et al. Timing and magnitude of increases in levothyroxine requirements during pregnancy in women with hypothyroidism. N Engl J Med2004;351(3): 241-9.
Articles and any other material published in the JAFES represent the work of the author(s) and should not be construed to reflect the opinions of the Editors or the Publisher.
Authors are required to accomplish, sign and submit scanned copies of the JAFES Declaration that the article represents original material that is not being considered for publication or has not been published or accepted for publication elsewhere.
Consent forms, as appropriate, have been secured for the publication of information about patients; otherwise, authors declared that all means have been exhausted for securing such consent.
The authors have signed disclosures that there are no financial or other relationships that might lead to a conflict of interest. All authors are required to submit Authorship Certifications that the manuscript has been read and approved by all authors, and that the requirements for authorship have been met by each author.