Globally, there are 387 million individuals living with type 2 diabetes, 46.3% of whom are undiagnosed.[1] A disproportionate diabetes burden is carried by the South East Asian and Western Pacific nations, as it affects 75 million people in South East Asia (8.3% of the adult population) and 138 million in the Western Pacific (8.5% of the adult population).[1] With this rising trend, it is estimated that Asia will contribute to more than 60% of the world’s diabetic population.[2] Furthermore, Asians have a strong ethnic and genetic predisposition for diabetes and lower thresholds for environmental risk factors.2 Within the Asian region, diabetes and pre-diabetes are more prevalent among Indians (37.9 and 18.9%, respectively) and Malays (23.8 and 22.6%, respectively). Pre-diabetes is more prevalent in women (21.9%) and urban dwellers (21.5%).[3]
Of particular concern is the evidence indicating that a significant proportion of individuals with diabetes or pre-diabetes are unaware of their condition. Consequently, adoption of risk reduction behavior is suboptimal. An evaluation of the United States National Health and Nutrition Examination Survey (NHANES 2005-2006) data revealed that almost 30% of the adult population had pre-diabetes but only 7.3% were aware of their condition.[4] Further, estimates for the prevalence of undiagnosed diabetes in South East Asia and the Western Pacific region were high (52.8 and 53.6%, respectively).[1] A cross-sectional survey of Malaysians found that the prevalence of newly diagnosed type 2 diabetes reached 12.6% adults, with an increasing prevalence of undiagnosed diabetes observed with age.[5] This high prevalence of undiagnosed diabetes poses a serious problem for public health, warranting an escalated effort to address and improve the burden of diabetes.[5]
METHODOLOGYThe consensus working group consisted of an assembly of 13 regional experts working as academics, researchers, clinicians, and policy makers from national and international organizations. The experts have substantial knowledge in relevant disciplines, including endocrinology and metabolism.
The expert group used a qualitative approach involving a question-and-answer format to shape and direct the flow of the discussions. This was followed by a comprehensive literature review of published academic articles for identifying the research evidence to guide recommendations. The research findings were subsequently triangulated and circulated electronically among all consensus group members.
The recommendations were formulated by the chairperson and members of the advisory board committee, after the initial group discussion and multiple e-mail communications. The diagnostic cut-off fasting plasma glucose (FPG), glycated haemoglobin (HbA1c), and oral glucose tolerance test (OGTT) values for pre-diabetes are based on the consensus-based outcomes of that meeting and the subsequent literature review.
Consensus was a priori defined as agreement of a large majority of advisory group members, without strong disagreements. If consensus was not reached, the working group would take a vote, where at least a simple majority vote would be required for the recommendation to pass. Any dissenting opinion would be captured and presented in the report. Every effort was made to achieve consensus among the committee members, and consensus was reached on every recommendation.
A preliminary review of the literature showed that several consensus statements exist, including the official position of the American Association of Clinical Endocrinologists and American College of Endocrinology, and another by the working group of the Spanish Diabetes Society.[6],[7] However, this consensus statement was written to reflect the various aspects of pre-diabetes management from an Asian perspective. Majority of the consensus statements were crafted based on studies conducted in Western populations without consideration of the cultural nuances that are sensitive and specific to Asian populations.
Recommendations for Pre-diabetes Management in Asian PatientsAt a regional consensus meeting held in Manila, Philippines on June 6, 2014 sponsored by Merck, experts from the Asia-Pacific region convened to develop consensus recommendations to guide clinicians in the management of Asian patients with pre-diabetes. The following are their recommendations:
Recommendation 1: Screening and diagnosis of pre-diabetesScreening and treatment of IGT can delay or prevent the development of type 2 diabetes, providing a window of opportunity for primary prevention of diabetes and CVD. The authors recommend targeted screening for patients aged ≥35 years and/or high risk individuals, followed by laboratory tests (i.e., FPG, HbA1c and/or 75-gram OGTT). High risk individuals include overweight or obese patients (country-specific), family history of diabetes, high blood pressure, dyslipidemia, history of large babies or gestational diabetes.
Pre-diabetes is diagnosed if the FPG ≥ 100 mg/dL (5.5 mmol/L), 2-hour OGTT is 140-199 mg/dL (7.8-11.0 mmol/L), and HbA1C of >5.7%. If initial screening of pre-diabetes is negative, patients should be rescreened every 1 to 3 years, depending on risk factors (based on clinical judgment) and local resource availability. The optimal cut-offs for diagnosing pre-diabetes in Chinese patients are HbA1c of 5.6% (38 mmol/mol) in the young and middle-aged and 5.7% (39 mmol/mol) in the elderly.[8]
The bases for the cut-off of 35 years and older for routine screening were reports from Bangladesh and Eastern Uganda that these individuals could have abnormal glucose regulation with a normal body mass index (BMI).[9],[10] Together with majority consensus voting, these formed the rationale for periodic screening of all persons older than 35 years for pre-diabetes. The HbA1c threshold set at 5.7% when screening for pre-diabetes was based on a previously published cost-effectiveness strategy.[11] Therefore, the authors recommend age-specific cut-offs for detecting pre-diabetes or diabetes in populations where such differences have been observed.
Recommendation 2: Treatment of pre-diabetesCurrent guidelines on diabetes prevention recommend intensive lifestyle intervention as the cornerstone of pre-diabetes management.[12],[13],[14],[15],[16],[17] The authors strongly recommend lifestyle intervention, preferably with a dietician referral specifically encompassing the following:
- Reduced intake of simple sugars
- Reduced fat intake, specifically saturated fats and oils
- Reduced trans fatty acid intake
- 5 to 10% weight loss from baseline
- Total calorie intake deficit based on target weight loss
- 30 minutes of exercise 5 to 7 times per week
Patients should ideally undergo a review after a period of 3 to 6 months.
Recommendation 3: Pharmacologic therapyPharmacologic intervention is recommended if there is inadequate response to lifestyle intervention after 3 to 6 months. Metformin should be initiated at a starting dose of 500 mg/day titrated up to a maximum of 2,000 mg/day as required. Alternative treatment should be considered if the patient is nonresponsive or intolerant to metformin (e.g., acarbose), or when it is contraindicated. Follow up is recommended at 3 to 6 months. These recommendations are in line with international and local guidelines and reflect current practice within the region.[12]-[15],[17] However, important considerations concerning pharmacotherapy with metformin for high-risk individuals should be emphasized, because the impact of duration of therapy with metformin, and long term cost-effectiveness of such early intervention, remain unclear.[18]
Burden Attributable to Pre-diabetes in AsiaIndividuals with impaired fasting glucose (IFG) and/or IGT are considered to have pre-diabetes, indicating a relatively high risk for future development of diabetes.19 IFG and IGT should not be viewed as clinical entities in their own right but rather as risk factors for diabetes as well as cardiovascular disease (CVD).[19] Both IFG and IGT are associated with obesity or adiposity (especially abdominal or visceral obesity), dyslipidemia with high triglycerides and/or low high density lipoprotein (HDL) cholesterol, and hypertension.
Increasing evidence shows that without targeted intervention, the progression from IGT to type 2 diabetes occurs more frequently in Asians compared with Caucasians.[20],[19],[19] The population-based controlled Da Qing study (n=110,660, aged 25 to 74 years), which explored the incidence of progression to diabetes among 577 IGT subjects, reported an incidence rate of 15.7% per 100 person-years in Chinese individuals with IGT.20 In Indians, the progression rate of pre-diabetes to diabetes was found to be as high as 18.3% per year.[19] These data are in stark contrast to Finnish (with an average progression rate of 6% per year) and American (11% per 100 person-years) individuals with pre-diabetes.[22],[23]
The prevalence of overweight/obesity (BMI ≥ 25 kg/m2) is increasing rapidly in the Asia Pacific region. It is important to note that a significant proportion of adults have undiagnosed diabetes, suggesting that the prevalence within the region is likely to be higher than is currently reported (Table 1).[1],[24] An IGT comparative prevalence in excess of 10% has been reported in Malaysia (15.19%), Hong Kong (13.30%), Japan (12.64%), Singapore (12.40%) and Taiwan (11.19%).[24]
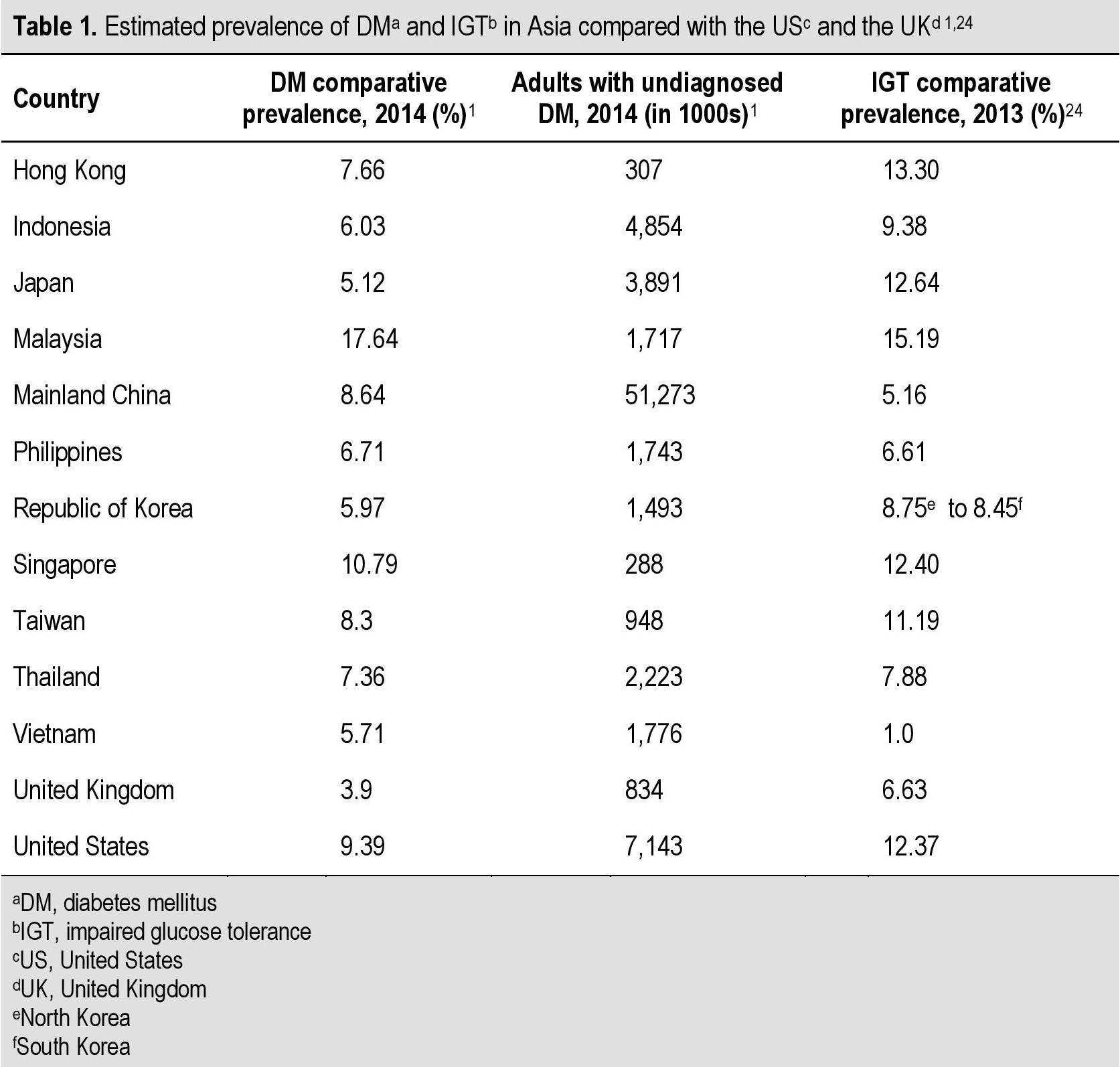
Table 1. Estimated prevalence of DMa and IGTb in Asia compared with the United States and the United Kingdom[1],[24]
In Thailand, it is estimated that 4.1 million adults have IGT.24 India is estimated to have 21.5 million adults with IGT, making it the country with the highest number of persons with diabetes in the South East Asian region.[24],[25] Almost one-third of adults with IGT are younger than 40 years of age. It is projected that by 2035, one in 8 adults will have IGT, while one in 10 will have diabetes. Therefore, prompt and effective IGT management is essential.[24] A similar pattern is emerging in the Philippines where the prevalence of pre-diabetes (i.e., combined IFG and IGT) was 31.3%, while it was 17.5% for IFG and 23.9% for IGT.[26]
Of note, the high prevalence of IGT throughout the region indicates that the incidence of diabetes is likely to increase over the coming decade. The alarming prevalence of pre-diabetes (IFG and/or IGT) and diabetes in the Asian region warrants urgent strategies aimed at preventing the conversion from pre-diabetes to diabetes.[27]
Current Management of Patients with Pre-diabetesNot everyone with IGT will subsequently develop type 2 diabetes. A large body of evidence supports the effectiveness of lifestyle interventions, specifically healthy diet and physical exercise, to prevent progression to diabetes.[20],[22],[23] The primary aim of lifestyle interventions is to prevent or delay the development of type 2 diabetes and its complications by targeting obesity and physical inactivity, which are the two most important modifiable risk factors of diabetes development.
The beneficial effect of lifestyle interventions has been confirmed in Asian populations. Among 577 Chinese individuals with IGT, long-term lifestyle interventions involving diet and/or exercise have been shown to significantly decrease the incidence of diabetes.[20] Over a 6-year period, there was a reduction in the incidence of diabetes in individuals practicing lifestyle interventions. Compared with the control group, both diet and exercise resulted to reductions in the risk of developing diabetes (31% and 46%, respectively).[20] These findings were replicated in the Finnish Diabetes Prevention Study (DPS) in 522 overweight (mean BMI ≥31 kg/m2) adults aged 40 to 65 years with IGT.22,28 At 4 years, the cumulative incidence of diabetes was 11% in the intervention group compared with 23% in the control group.[22] From these results the authors concluded that one case of diabetes could be prevented by treating 22 IGT patients with lifestyle intervention for one year, or 5 patients for a period of 5 years.[22] The Diabetes Prevention Program (DPP) in the US, one of the largest randomized controlled clinical trials to date, found similar results. In a population of 3234 adults with IGT and/or IFG (with a mean BMI of 34.0 kg/m2 and a mean age of 51 years), the incidence of diabetes for the lifestyle intervention group was lower than in the placebo group (4.8 and 11.0 cases per 100 person-years, respectively).[23] The authors concluded that one case of diabetes could be prevented by treating 7 patients with intensive lifestyle modification for 3 years.23 These studies found that type 2 diabetes can be prevented by lifestyle changes in those deemed to be at high-risk, such as those with IGT (Table 2).[20],[22],[23]
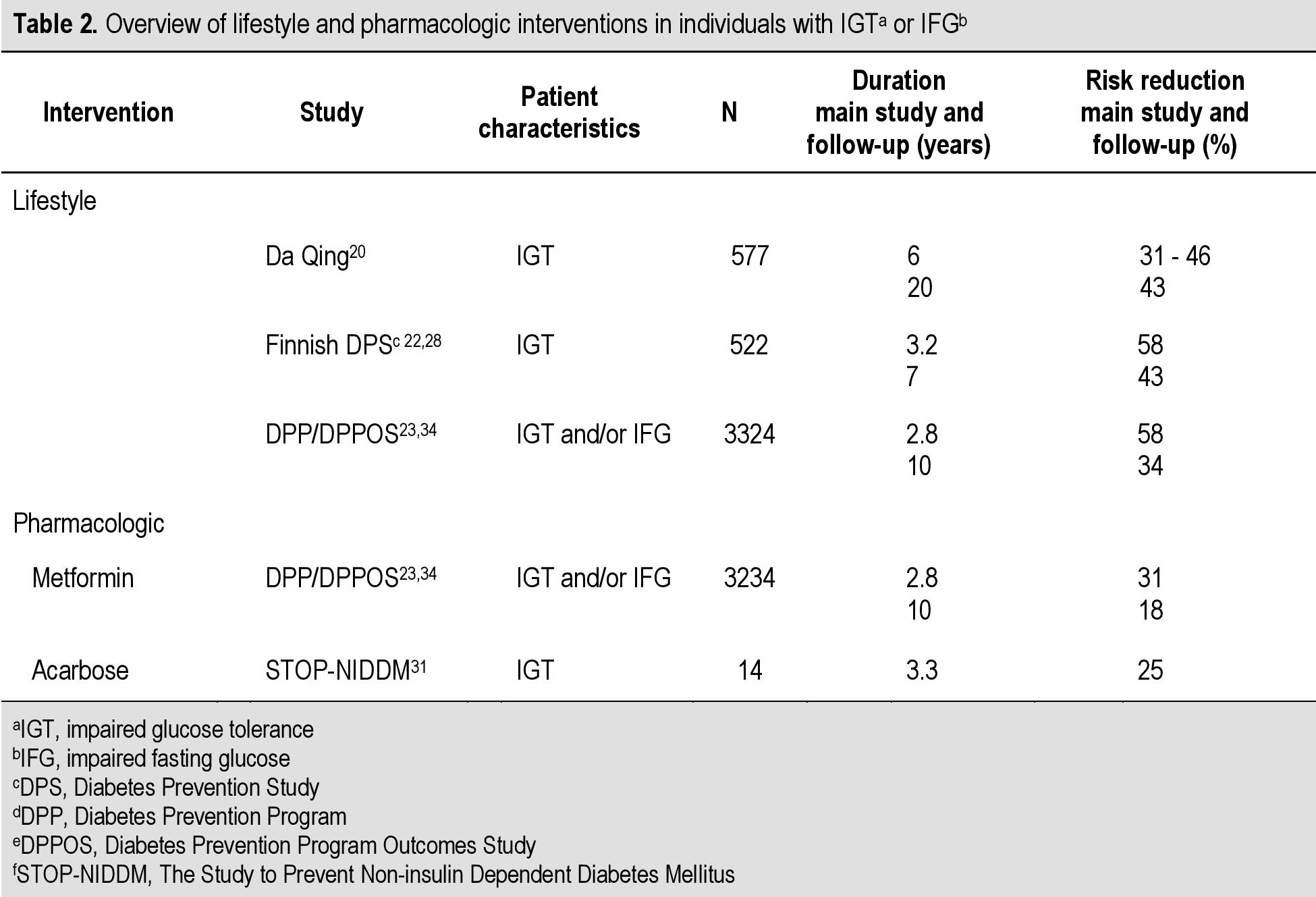
Table 2. Overview of lifestyle and pharmacologic interventions in individuals with IGT [Impaired Glucise Tolerance] or IFG [Impaired Fasting Glucose][1],[24]
The DPP study also showed that treatment with metformin could also delay or prevent type 2 diabetes. Compared with placebo, metformin reduced the incidence of type 2 diabetes by 31% (Table 2).[23] A subsequent washout study showed that approximately one-quarter of this effect could be accounted for by the pharmacologic effect of metformin that disappeared following discontinuation of the therapy.[29] Notably, even after the washout period, a significant 25% reduction in the incidence of diabetes persisted.[29] Although the DPP study was not powered to demonstrate statistical significance for between subgroup effects, analysis revealed that metformin was more effective in patients with higher fasting plasma glucose (FPG) levels (≥ 110 mg/dL), in those younger than 60 years of age, and in individuals with a BMI ≥35 kg/m2.[23]
Despite the benefits of delaying or preventing the onset of diabetes, it has been shown that metformin is rarely prescribed as preventive therapy in working-age adults with pre-diabetes. Over a period of 3 years, only a minority of US adults with pre-diabetes (3.7%, or one in 27) were prescribed metformin, and only 7.8% (fewer than one in 12) of high-risk patients as identified by the national guidelines received metformin in a retrospective cohort study.[30] These findings highlight the need for intensive lifestyle modification programs. Patients should, at a minimum, be educated on the benefits of metformin and should ideally also be offered this option as preventive treatment for diabetes.
Acarbose has also been shown to effectively reduce the risk of progression to diabetes in individuals with IGT (Table 2).[31] The STOP-NIDDM trial reported a decrease in progression to diabetes by 25% of patients with IGT.[31] Patients on acarbose additionally improved in their tolerance to glucose as the probability of reversing to normal glucose tolerance was significantly higher in these patients than in those on placebo (p<0.0001).[31]
Positions on Specific Questions Addressed to Guide Recommendations
1. Do prevention interventions have sustained effects?A 20-year follow-up of the Da Qing study determined that combined lifestyle intervention resulted in a 51% reduction in the incidence of diabetes during active intervention and a 43% reduction over 20 years (Table 2).[32] The benefits of the active lifestyle intervention translated to an average delay of diabetes onset of 3.6 years..[32] The Da Qing study found that the 20-year cumulative diabetes incidence was 93% in the controls versus 80% among those who received the combined lifestyle intervention..[32] The 20-year follow-up did not detect significant differences in the incidence of first CVD events, CVD mortality, or all-cause mortality between the combined lifestyle intervention and control group, as it was not powered to detect statistical differences in these outcomes..[32]Nevertheless, the follow-up study showed that lifestyle interventions over 6 years were able to delay or prevent diabetes onset for up to 14 years after active intervention ceased..[32]
The most recent 23-year follow-up of the Da Qing study confirmed that active lifestyle intervention significantly reduced the risk of CVD and all-cause mortality. The cumulative incidence of CVD mortality was 11.9% for patients in the lifestyle intervention group and 19.6% in the control group. In terms of all-cause mortality, the cumulative incidence was 28.1% in the lifestyle intervention and 38.4% in the control group.[33] The significant differences in the incidence of diabetes between the two groups persisted during the 23-year follow-up: the cumulative incidence was 72.6% in the intervention group and 89.9% in the control group.[33] These findings justify the adoption of lifestyle interventions in patients with IGT.
2. Are we preventing type 2 diabetes or delaying it?Interventions that may prevent or delay IGT, which is associated with cardiovascular disease and conversion to type 2 diabetes, are clinically important. The 10-year follow-up of the DPP, the Diabetes Prevention Program Outcomes Study (DPPOS), found that patients who were on intensive lifestyle had a 34% reduction in the incidence rate of diabetes, with an average delay of diabetes progression by about 4 years versus placebo (Table 2).[34] Those treated with metformin had an 18% reduction in diabetes incidence rate, with the onset of diabetes delayed by an average of 2 years.[34]
It is clear that high risk patients must be identified and lifestyle changes should be implemented and sustained over the long term. Delaying or preventing type 2 diabetes is cost-effective and will help turn the tide in the diabetes epidemic.[35]
3. What is the current management of pre-diabetes in countries of the Asia-Pacific region?Most countries within the region do not have country-specific guidelines and therefore follow the American Diabetes Association (ADA) or the International Diabetes Federation (IDF) recommendations.[12],[13] The IDF consensus guidelines on the prevention of type 2 diabetes recommend the following three steps for the prevention of diabetes development: (1) identification of individuals at high risk of developing diabetes, (2) assessment of risk levels by measuring plasma glucose levels, and (3) initiation of lifestyle interventions with or without pharmacologic therapy.[36] Once individuals with pre-diabetes have been identified, they are advised to undergo structured lifestyle modifications, with the aim of achieving gradual and sustained weight loss and maintaining a healthy body composition through physical activity and change of dietary habits.[36] The World Health Organization (WHO) and IDF also recommend to address other risk factors, including smoking.[13],[37] In addition, the WHO highlights the need for a global approach to reduce the growing global burden of diabetes.[37]
The ADA recommends the referral of patients with IGT, IFG or HbA1c of 5.7 to 6.4% to an ongoing support program targeting weight loss of 7% of body weight and moderate exercise of ≥150 minutes per week.[12] The ADA states that metformin therapy for prevention of type 2 diabetes may be considered in individuals with IGT, IFG or an HbA1c of 5.7 to 6.4%, especially for those with BMI >35 kg/m2, individuals aged < 60 years, and women with prior gestational diabetes.[12] In addition, the ADA guidelines encompass recommendations for follow-up counselling for successful lifestyle interventions, annual monitoring of individuals with pre-diabetes for development of diabetes, as well as screening for and treating of modifiable risk factors for CVD.[12]
In Asia-Pacific countries, lifestyle modifications remain the mainstay of recommended first-line interventions for patients with pre-diabetes. In Malaysia, lifestyle interventions, such as diet and physical therapy, are the pillars for pre-diabetes therapy.[14] In addition, the 2015 Ministry of Health (MOH) guidelines state that metformin (as the preferred first-line oral anti-diabetic agent) should be considered for patients at very high risk of progressing to diabetes (combined IFG and IGT, IGT plus other risk factors, or failed lifestyle intervention after 6 months).[14] Off-label metformin may be initiated at the discretion of the prescribing physician.
In Thailand, there are no available guidelines for the management of pre-diabetes. Majority of physicians follow the IDF recommendations, ADA guidelines or findings from randomized controlled trials on pre-diabetes prevention.[12],[13],[20],[22],[23],[34] Likewise, given a lack of guidelines in the Philippines, physicians generally follow the ADA recommendations. However, in the Philippines, metformin is approved for the treatment of pre-diabetes after failed lifestyle intervention.
Guidelines in Singapore indicate that lifestyle modification should be the first-line treatment of choice.15 Metformin may be considered for individuals with a very high risk of progressing to diabetes, particularly patients with IFG, IGT, less than 60 years of age, or BMI ≥35kg/m2.[15] Hong Kong has pre-diabetes management guidelines aimed at the primary care sector.[16] The emphasis is mainly on lifestyle modifications using dietary or behavioral interventions to reduce and maintain body weight and practice healthy lifestyle. Pharmacologic therapy is not routinely recommended at present.[16]
The Indonesian guidebook on the management of pre-diabetes and prevention of type 2 diabetes 2009 [Buku Panduan - Pengurus Besar Persatuan Diabetes Indonesia (PB Persadia)] states that the diabetes prevention strategy should encompass a three-step process that includes identification of high-risk individuals (step 1), risk calculation, (step 2) and intervention (step 3).[17] Step 3 involves lifestyle changes, body weight management (reduction by 5 to 7% of baseline body weight, 0.5 to1 kg/week), physical activity and pharmacologic intervention. The latter involves either metformin given 250 to 850 mg twice daily in individuals 60 years or younger, with BMI >25 kg/m2 and FBS >110 mg/dL (6.1 mmol/L) if no contraindications are present; or acarbose 50 to 100 mg thrice daily.[17]
Within the region, despite the lack of formal recommendations, metformin is often used off-label for certain patient populations. For example, there are no pre-diabetes guidelines in India, but metformin is used off-label by physicians if pre-diabetes patients require pharmacotherapy. Pakistan also has no formal guidelines for pre-diabetes treatment. In view of long-standing safety information about metformin, this drug is prescribed to individuals who are noncompliant with lifestyle interventions. For other potential drugs, further long-term studies are needed on safety and vascular outcomes before lifelong treatment can be safely recommended.
Pre-diabetes represents a window of opportunity to prevent or delay the progression to diabetes and its associated complications, underscoring the critical need for screening at the primary care level. Lifestyle modification including weight loss, dietary changes and increased physical activity play a major role in controlling the disease. Furthermore, significant evidence support the effectiveness of combining lifestyle modification and pharmacologic therapy on certain patient populations in delaying the onset of diabetes. A cost-effectiveness analysis of lifestyle intervention and metformin therapy for the prevention of diabetes in Singapore concluded that both lifestyle modification and metformin are likely to be cost-effective and worth implementing in Singapore to prevent or delay the onset of type 2 diabetes.[35] Although the importance of lifestyle interventions is well recognized throughout Asia, many countries do not have formal recommendations to guide the diagnosis and management of individuals at risk of progression to diabetes.
Overall, these consensus recommendations provide a clear and concise approach to the management of individuals with IGT based on the available evidence and current best clinical practice. Furthermore, local applicability of these recommendations will be far-reaching, particularly in guiding action and policy for pre-diabetes and other related endocrine and metabolic disorders at the regional, national and local levels.
AcknowledgmentsThe authors would like to thank Merck Pte Ltd (Singapore) for providing an educational grant in support of the consensus meeting. Meeting and editorial support was provided by MIMS Pte Ltd through this grant.
Statement of AuthorshipAll authors certified fulfillment of ICMJE authorship criteria.
Author DisclosureThe authors have received honoraria for their participation in the APAC Regional Pre-Diabetes Advisory Board Meeting 2014. Professor Mirasol is a advisory board and speaker for AstraZeneca, Merck, Novo, Genzyme, Lilly, MSD.
Funding SourceMerck Pte Ltd (Singapore) provided logistic support and funding for the consensus meeting. The authors received fair market honorarium for their time spent attending the consensus meeting and developing the manuscript. Merck Pte Ltd (Singapore) also paid for MIMS Pte Ltd to provide editorial support. RM, ACT, AA, KT, CD, MM, MRS, BKS, SS, NNS, SS, RTT and FU reported personal fees from Merck Pte Ltd (Singapore) during the preparation of this manuscript.
[1] International Diabetes Federation. Diabetes Atlas 6th edition poster update, 2014. http://www.idf.org/sites/default/files/Atlas-poster-2014_EN.pdf. Accessed September 22, 2016.